Licensing Opportunity
Patent
Chemo-enzymatic site-specific modification of peptides and proteins to form cleavable conjugates
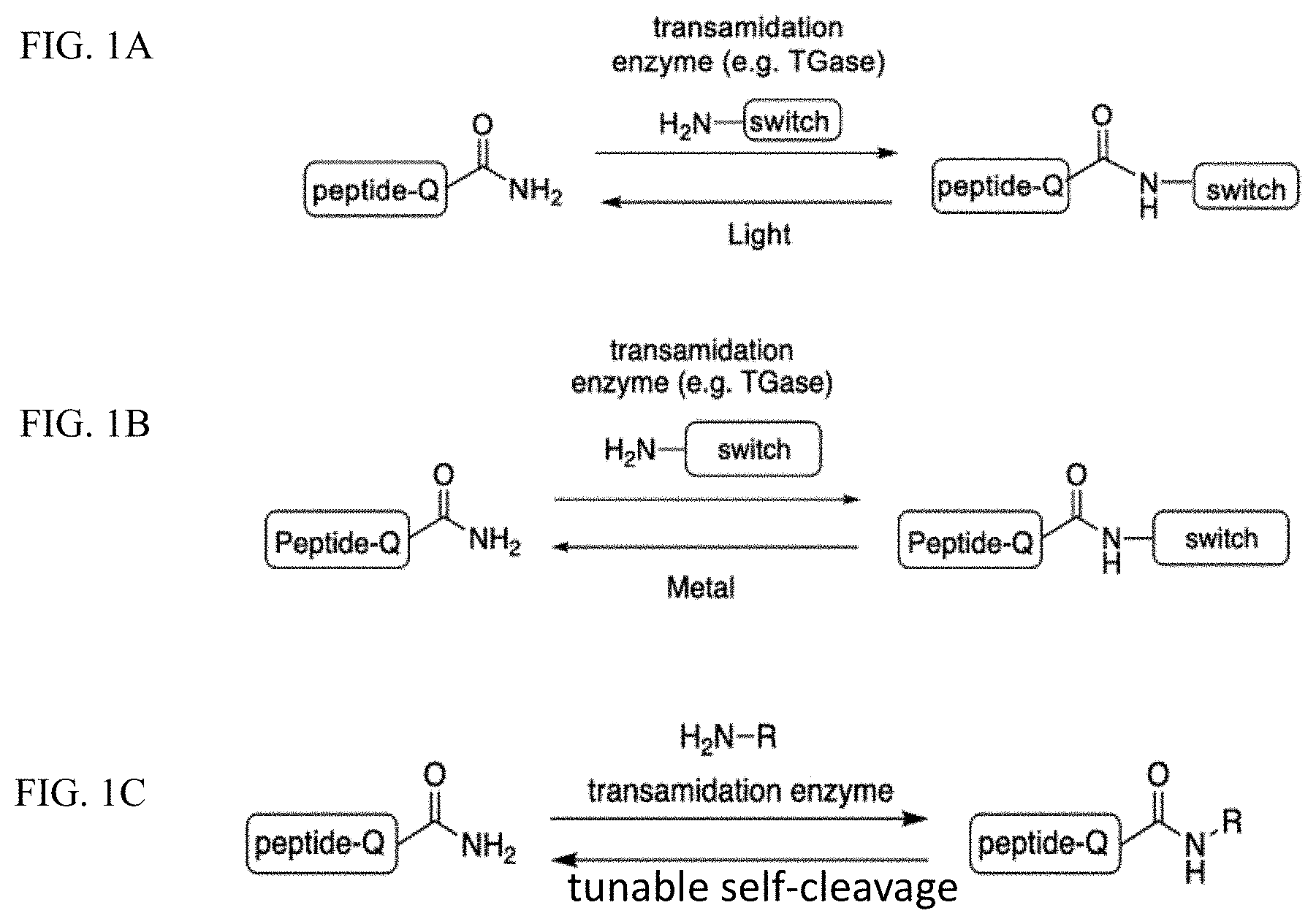
A method is provided for reversibly modifying a protein or peptide on its glutamine residue(s) by performing a reaction, such as a transglutaminase-catalyzed reaction, between the protein or peptide and an amine-containing reagent, whereby the reagent is linked through its amine function to a side chain of the glutamine residue. Subjecting the modified protein to an appropriate stimulus regenerates the protein or peptide in its original form.
Related Papers
.
Site-Specific Reversible Protein and Peptide Modification: Transglutaminase-Catalyzed Glutamine
Conjugation and Bioorthogonal Light-Mediated Removal
.
Dynamic photoswitches in proteins that impart spatial and temporal control are important to manipulate and study biotic and abiotic processes. Nonetheless, approaches to install these switches into proteins site-specifically are limited. Herein we describe a novel site-specific method to generate photoremovable protein conjugates. Amine-containing chromophores (e.g., venerable o-nitrobenzyl and less-explored o-nitrophenylethyl groups) were incorporated via transamidation into a glutamine side chain of α-gliadin, LCMV, and TAT peptides, as well as β-casein and UmuD proteins by transglutaminase (TGase, EC 2.3.2.13). Subsequently, photolysis regenerated the native peptides and proteins. When this modification leads to the reduction or abolishment of certain activities, the process is referred to as caging, as in the case for E. coli polymerase manager protein UmuD. Importantly, this method is simple, robust, and easily adaptable, e.g., all components are commercially available.
Provisional Patent
.
For more information of licensing and contracts please see, Center for Research Innovation at Northeastern University.